PRODUCT INSIGHTS
SINGLE INCISION STRESS URINARY INCOTINENCE TREATMENT
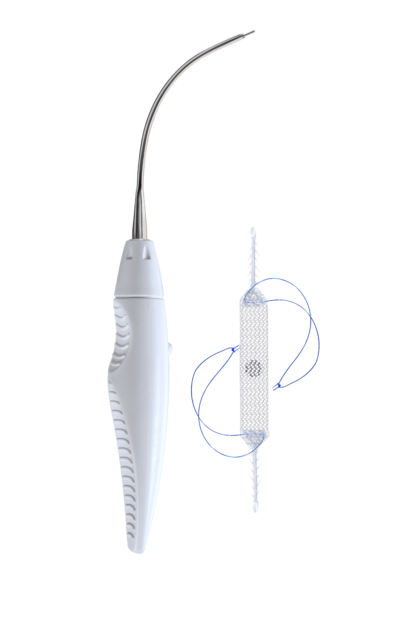
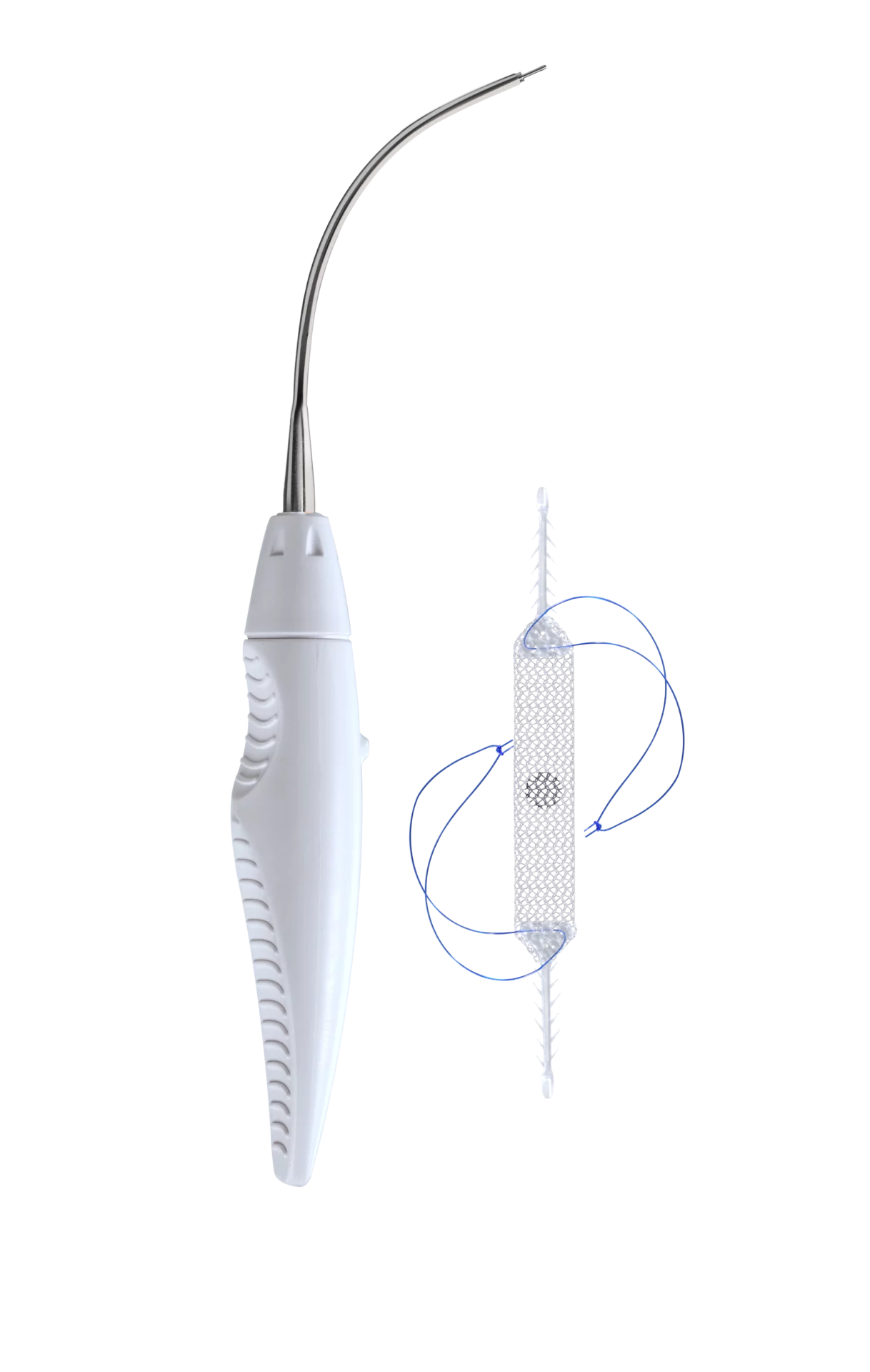
The Ophira sling is intended to be used for the treatment of female stress urinary incontinence resulting from urethral hypermobility and/or intrinsic sphincter deficiency1. The Mini Sling with its two fixations arms on each side, is fixated to both internal obturator muscles and allows to create support underneath the urethra while using only one incision for the implantation.
CONSISTENT CLINICAL RESULTS
Clinical evidence obtained with Ophira Mini Sling System is consistent in different centers around the world. Ophira offers a safe and efficient method for the treatment of female stress urinary incontinence2. Compared to transobturator and retropubic suburethral slings, Ophira shows no inferiority regarding success rate at mid term follow-up3,4,5,6,7. The major advantage is the possibility of performing this procedure under regional or local anaesthesia5. The vaginal sling application of Ophira led to a further reduction of complications by avoiding the retropubic or transobturator space6. Its unique fixation system results in immediate high efficacy8.
- Ophira Instructions for Use 2023
- Yildiz G, Ceylan Y, Ucer O, Arslan D, Çelik O, Gunlusoy B. Safety and efficacy of single-incision sling for female stress urinary incontinence: 3 years‘ results. Int Urogynecol J. 2016 Mar 18.
- Smith P.P. et. Al: Comparison of single-incision mid-urethral tape (OphiraTM) and transobturator tape (ObtryxTM) sub-urethral sling procedures for female stress urinary incontinence. Journal of Clinical Medicine and Research. Vol. 5(4), pp. 53-57, June, 2013. DOI 10.5897/JCMR12.019. (2013)
- Jurakova M, Huser M, Belkov I, et al. Prospective randomized comparison of the transobturator mid-urethral sling with the single-incision sling among women with stress urinary incontinence: 1-year follow-up study. Int Urogynecol J. 2016;27(5):791-796. doi:10.1007/s00192-015-2895-2
- Sharifiaghdas F, Nasiri M, Mirzaei M, Narouie B. Mini Sling (Ophira) versus Pubovaginal Sling for Treatment of Stress Urinary Incontinence: A Medium-term Follow- up. Prague Med Rep. 2015;116(3):210-218. doi:10.14712/23362936.2015.60
- Golbasi C, Taner CE, Golbasi H. Long-term outcomes and quality of life effects of single incision mini sling procedure in stress urinary incontinence patients. Eur J Obstet Gynecol Reprod Biol. 2019;234:10-13. doi:10.1016/j.ejogrb.2018.12.0307.
- Djehdian LM, Araujo MP, Takano CC, Del-Roy CA, Sartori MG, Girão MJ, Castro RA. Transobturator sling compared with single-incision mini-sling for the treatment of stress urinary incontinence: a randomized controlled trial. Obstet Gynecol. 2014 Mar;123(3):553-61. doi: 10.1097/AOG.0000000000000148
- Palma P, Riccetto C, Bronzatto E, Castro R, Altuna S. What is the best indication for single-incision Ophira Mini Sling? Insights from a 2-year follow-up international multicentric study. Int Urogynecol J. 2014;25(5):637-643. doi:10.1007/s00192-013-2242-4.
PRODUCT HIGHLIGHTS
OPHIRA MINISLING IMPLANT
Ophira, with its Type 1 polypropylene mesh and its thermosealed edges, offers an excellent balance of the main biomechanical requirements:
tissue integration and low elasticity.
OPHIRA FIXATION SYSTEM
Ophira’s fixation system has an unique and innovative design with multiple fixation points along its self-fixating arms. It offers a high pull out force and reliability in its primary fixation. A reliable primary fixation is important for the outcome of the treatment. The Ophira Minisling System offers a safe and simple way to achieve a correct and precise sling placement3.
OPHIRA INSERTION GUIDE
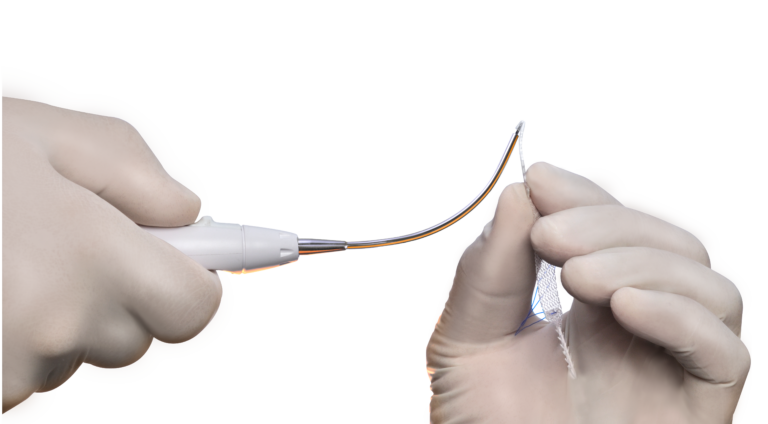
The Retractable Insertion Guide (RIG) is designed to provide control and easy handling that allows a precise and minimally invasive implantation technique. The anti-rotational assembly system on the tip of the retractable insertion guide goes along with the anti-rotational connector of the fixation arm and ensures that the minisling can be implanted without positioning complications. With its 2.2 mm diameter and anatomical curvature the RIG enables the surgeon to implant the fixation arms safe and efficient in the right anatomical direction, while having a very short blind passage.
SURGICAL TECHNIQUE
OPHIRA IN DAILY CLINICAL PRACTICE
- Intention for use
- 15.000+ Ophira Minislings implanted
SURGICAL TECHNIQUE
OPHIRA IN DAILY CLINICAL PRACTICE
- Intention for use
- 15.000+ Ophira Minislings implanted
CONTRAINDICATIONS &
PRECAUTIONS
Ophira should be used only by surgeons familiar with the procedures and techniques for implanting Ophira. Careful patient selection and complete diagnostic evaluation are essential prior to surgery.
Ophira should not be prescribed if there are infectious processes, especially in the genital system or in the urinary tract.
OPHIRA should not be used for PATIENTS:
- on anticoagulant therapy
- with current urinary infection or,
- during pregnancy,
- with known sensitivity or allergy to polypropylene products,
- with pre-existing conditions that pose an unacceptable surgical risk.
In order to avoid complications, please consider all warnings and precautions, which are stated in the IFU.


