
Taking the artificial
urinary sphincter concept
to the next level
PRODUCT INSIGHTS
TREATMENT FOR MALE STRESS URINARY INCONTINENCE
VICTO is an adjustable artificial urinary sphincter, and it is intended to restore the natural process of urinary control, simulating normal sphincter function by opening and closing the urethra under the patient’s will1.
It is indicated for the treatment of male stress urinary incontinence caused by intrinsic sphincter deficiency (ISD) in cases such as post-prostatectomy incontinence1.
5 YEARS WARRANTY
- Victo Instructions for Use 2022.
PRODUCT HIGHLIGHTS
You are currently viewing a placeholder content from Default. To access the actual content, click the button below. Please note that doing so will share data with third-party providers.
More InformationI’m interactive - move and click me!
Too small? Please click here
CONFIGURATION
This pre-connected artificial urinary sphincter, consist of a control pump, a pressure regulating balloon (PRB) and a urethral cuff. VICTO was developed to restore urinary continence by keeping the urethra closed by means of the cuff that exerts pressure around the urethra, which is regulated by the PRB.
By pressing the control pump placed inside the scrotum, the cuff deflates, thus opening the urethra to enable urination. The urethra is then occluded automatically after a couple of minutes.

ADJUSTABLE DEVICE
The device enables pressure adjustments postoperatively, through the self-sealing titanium port in the pump, in an outpatient and minimally invasive procedure. VICTO can be set to the lowest base pressure necessary to restore continence.

MULTIPLE CUFF SIZES
Available in different cuff-sizes, to fit the entire range required for different urethral sizes of every patient.
SURGICAL TECHNIQUE
SURGICAL TECHNIQUE
DEACTIVATION PROCEDURE
If deactivation of the VICTO is necessary, this should be done by removing 7 ml of fluid from the system. For this purpose, adequate skin disinfection is mandatory prior to percutaneous puncture of the port.
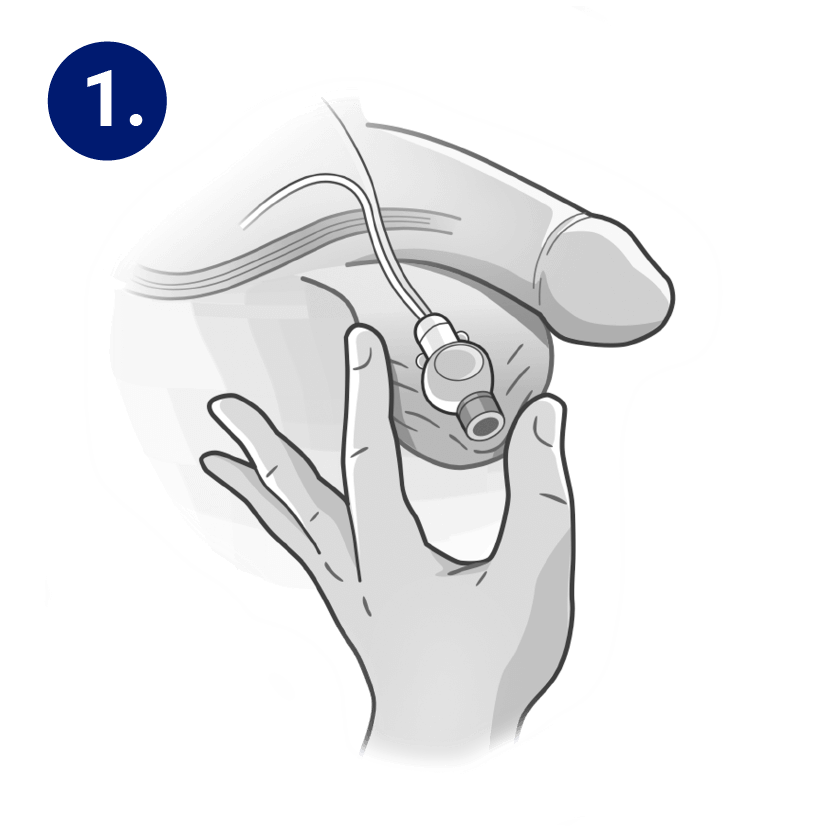
STEP 1
Using clean gloves, palpate and identify the control pump through the scrotal skin. Scrotal skin should be cleaned with antiseptic substance to minimize risk of infection.
Connect a non-coring needle 25G x 5/8” to a 10ml Luer-Lock syringe.

STEP 2
Hold the pump firmly in place and insert the non-coring needle into the access port until it contacts the titanium protection cup. The insertion of the needle should be perpendicular and concentric to the port.
Precautions should be taken when inserting the needle, so as not to puncture the walls of the pump other than the self-sealing port.

STEP 3
Extract 7ml from the VICTO by pulling the plunge of the syringe and remove the needle from the port of the pump.
After performing the deactivation process, the VICTO sphincter will no longer provide pressure around the urethra. To restore its normal functioning after the patient´s medical intervention has been completed, 7ml should be reinjected into the VICTO.
DEACTIVATION PROCEDURE DE DEACTIVATION PROCEDURE ENCONTRAINDICATIONS &
PRECAUTIONS
The main contraindications for the implantation of VICTO are listed as follows:
- Uncontrolled overactive bladder
- Untreated or irreversible intravesical obstruction
- Untreated acute urinary tract infection, active infection at the surgical sites or systemic infections (sepsis)
- Patients with stricture/stenotic disease
- Known allergy to silicone or titanium
- Impaired cognitive or manual abilities which may affect manual dexterity or motivation, and prevent the patient from operating the device
- Patients whom the surgeon determines to be not suitable due to risks associated with open surgical procedures and/or with the patients’ medical history (physical or mental problems).
- Prior abdominal surgery, status post peritonitis, and other conditions resulting in peritoneal adhesions, fibrosis, scars that could potentially interfere with the correct functioning of the pressure regulating balloon.
EMEA / APAC
| DESCRIPTION | CUFF SIZE | ORDERING CODE |
|---|---|---|
| VICTO STARTER KIT 3.7 |
3.7 | KIT-VICTO-37 |
| VICTO STARTER KIT 4.0 |
4.0 | KIT-VICTO-40 |
| VICTO STARTER KIT 4.5 | 4.5 | KIT-VICTO-45 |
| VICTO STARTER KIT 5.0 | 5.0 | KIT-VICTO-50 |
| VICTO STARTER KIT 5.5 |
5.5 | KIT-VICTO-55 |
| DESCRIPTION | ORDERING CODE |
|---|---|
| ACCESSORIES KIT | KIT-AC-02 |
LATAM
| DESCRIPTION | CUFF SIZE | ORDERING CODE |
|---|---|---|
| VICTO STARTER KIT 3.7 |
3.7 | KIT-AUS-37-WS |
| VICTO STARTER KIT 4.0 |
4.0 | KIT-AUS-40-WS |
| VICTO STARTER KIT 4.5 | 4.5 | KIT-AUS-45-WS |
| VICTO STARTER KIT 5.0 | 5.0 | KIT-AUS-50-WS |
| VICTO STARTER KIT 5.5 | 5.5 | KIT-AUS-55-WS |
| DESCRIPTION | ORDERING CODE |
|---|---|
| ACCESSORIES KIT | KIT-AC-01 |
| DESCRIPTION | ORDERING CODE |
|---|---|
| ADJUSTMENT KIT | KIT-AD-06 |
| DESCRIPTION | ORDERING CODE |
|---|---|
| VICTO PUMP | KIT-RP-03-10-06 |
| VICTO BALLOON | KIT-RB-01 |
| VICTO CUFF 3.7 | KIT-RC-37-10-06 |
| VICTO CUFF 4.0 | KIT-RC-40-10-06 |
| VICTO CUFF 4.5 | KIT-RC-45-10-06 |
| VICTO CUFF 5.0 | KIT-RC-50-10-06 |
| VICTO CUFF 5.5 | KIT-RC-5.5-10-06 |
All replacement kits are not available in BRAZIL at the moment.


