
PRODUCT INSIGHTS
ENDOSCOPIC TREATMENT OF VESICOURETERAL REFLUX
VANTRIS is a biocompatible, non-absorbable, synthetic tissue-bulking substance, for the endoscopic treatment of vesicoureteral reflux (VUR) in pediatric population1. VANTRIS consists of particles of polyacrylate polyalcohol copolymer (PPC) immersed in a carrier and it is presented in a 1ml syringe, ready to be easily and manually injected with 22 or 23-gauge needles via suburethral injection procedure (STING technique)2.
HIGH SUCCESS RATE
VANTRIS has a median success rate of 91,55% of patients, most of the cases after a single injection. It has also proven to be effective even on high-grade reflux patients.


- VANTRIS Intructions for use
- Promedon files
PRODUCT HIGHLIGHTS
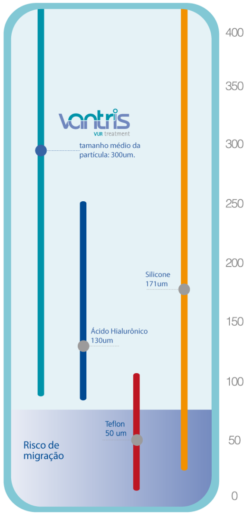
SAFE MACROPARTICLES
VANTRIS biocompatible and synthetic particles have an average size of 300um (min-max: 90um-1000um) which prevents the risk of migration from the implantation site.
STABLE BULKS
The non-biodegradable nature of VANTRIS gives high stability and durability to the bulk beneath the ureteral orifice, providing long-term effectiveness.
MINIMALLY INVASIVE PROCEDURE
The endoscopic treatment to implant VANTRIS is minimally invasive procedure and may be performed on an outpatient basis. This allows patients to return to their normal daily activities after a short period of time.

EASY TO IMPLANT
VANTRIS particles are amorphous and highly deformable by compression, and these characteristics guarantee the fluidity of the hydrogel. The material can be extruded by 22 and 23 G needles, allowing optimal injection precision with minimum tissue damage.

CONTRAINDICATIONS &
PRECAUTIONS
The main contraindications are:
- Ureterocele
- Voiding dysfunction
- Kidney dysfunction
- Paraureteral (Hutch) diverticulum
- Untreated or active urinary tract infections
- Ureterovesical junction obstruction
- Known allergy/ hypersensitivity to the device materials
HELPFUL RESOURCES
ORDERING INFORMATION
| PRODUCT | ORDERING CODE |
|---|---|
| 1ml VANTRIS SYRINGE | BAR-1J |
| DESCRIPTION | LENGTH | NEEDLE TIP | ORDERING CODE |
|---|---|---|---|
| SEMI-RIGID BEVEL TIP | 350mm | 22G | RINS |
| SEMI-RIGID SIDE OPENING | 350mm | 22G | RIN |
| 5.0Fr FLEXIBLE NEEDLE | 350mm | 23G | 50F |
| 3.7Fr FLEXIBLE NEEDLE | 350mm | 23G | 37F |