PRODUCT INSIGHTS
RETROPUBIC SLINGS SYSTEM TO TREAT FEMALE STRESS URINARY INCOTINENCE
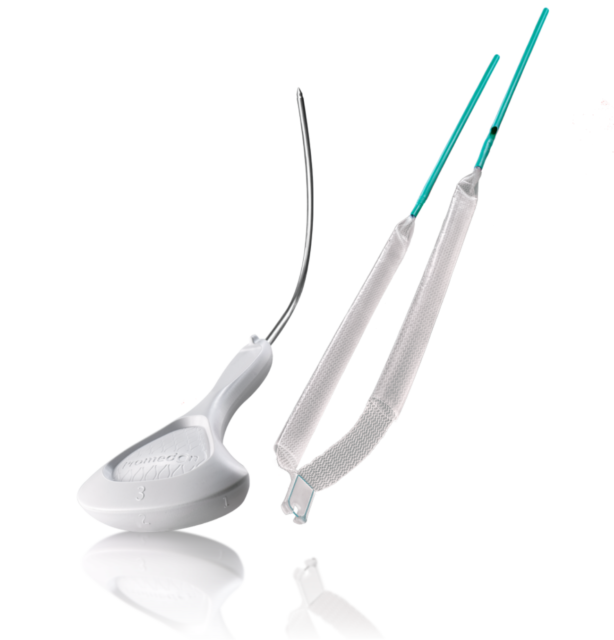
STEEMA Retro is a retropubic sling system, developed for the treatment of female urinary incontinence. The transvaginal sling system, equipped with green connector arms on each side, the Teflon® sheath and the positioning tab, brings all features which helping the surgeon to perform an easy, secure and gentle implantation. Once the implantation is completed, only the sling made from polypropylene type 1 is left in the patient to give support to the urethra and therefore restore continence. Right after the implantation, before tissue ingrowth, the sling anchors itself to the tissue by using a Velcro®-like effect and gets along without additional fixation.
PRODUCT HIGHLIGHTS
STEEMA RETROBUBIC SLING
Steema is a Premium Retropubic Sling, designed to provide surgeons with a better and safer procedure. Beside the features which supports the surgeon within the implantation, the sling made from polypropylene typ 1 with its thermosealed edges, offers an excellent balance of the main biomechanical requirements, as tissue integration and low elasticity, to enable the restorage of continence.
STEEMA RETROBUBIC NEEDLE
The STEEMA Retropubic Needle and the green connector arms form a reliable team to lead the sling through the retropubic space smooth and safely.
RETROPUBIC SLING SYSTEM COMPARISON
| FEATURE | GYNECARE TVT EXACT SYSTEM | ADVANTAGE FIT SYSTEM | STEEMA RETROPUBIC SLING SYSTEM | KEY DIFFERENTIATOR |
|---|---|---|---|---|
| Needle Radius | 106.5 mm | 82.5 mm | 106.2 mm | Designed to properly embrace the pubic bone and reduce the possibility of injury. |
| Connector arms color | White | Blue | Green | Guarantee the clearest contrast during cytoscopy. |
| Position Tab | No | Yes | Yes | Provides a controlled and easy sling positioning. |
| Sling Positioning Force | 15.8 N | 17.3 N | 12.1 N | Designed to provide a smooth passage during the sling positioning. |
| Sling Elasticity Index | 3.7 mm/N | 11.1 mm/N | 2.9 mm/N | High resistance to deformation avoiding potential urethral damage. |
SURGICAL TECHNIQUE
STEEMA RETRO IN DAILY CLINICAL PRACTICE
STEEMA Retropubic has been designed for the treatment of female stress urinary incontinence
SURGICAL TECHNIQUE
STEEMA RETRO IN DAILY CLINICAL PRACTICE
STEEMA Retropubic has been designed for the treatment of female stress urinary incontinence
CONTRAINDICATIONS &
PRECAUTIONS
STEEMA Retropubic should not be prescribed if there is any type of infection, especially genital or related to the urinary tract.
STEEMA Retropubic should not be used in patients who:
- are pregnant
- are sensitive or allergic to polypropylene products,
- have pre-existing pathologies or conditions posing an unacceptable surgical risk,
- have soft tissues pathologies in the site intended for implant placement, or
- have pathologies or other conditions that compromise wound healing.
For further precautions and warnings, we refer to our Instruction for Use




